Vial InspectionBecause Health Matters
Maximum Product Safety in the Inspection of Vials and Seals
360°-Sensor for Crimp Quality Inspection
Vial Inspection
Vials and Crimping—Fully Automated Monitoring of Production Quality
Patient safety begins during the manufacturing of primary packaging for pharmaceutical products. It is important to be able to depend on technology that monitors the process chain in a reliable and cost-effective manner. Which is exactly what automated visual inspection (AVI) does. The result? Maximum product safety, cost reductions, and rapid profitability!
Tackle Defects at the Source
Pharmaceutical products and their primary containers must meet rigorous quality standards. During vial filling and packaging, a very large number of production steps must be carried out under strict hygiene conditions.
AVI inspects along the complete process chain—from pre-filling to filling, closing, and packaging. Defects are detected at their point of origin.

False Rejects—No Thanks!
Defective vials and crimp caps are filtered out at an early stage to prevent them reaching further processing. This is an important cost factor for you, as it prevents defective containers from being filled with costly pharmaceuticals.
With AVI, high-powered sensors and intelligent algorithms reliably detect defects and keep false rejects to a minimum. But that’s not all. You can also use the inspection data collected during AVI for analysis and process optimization.
- < 0,3%typical false reject rate
100% Process Control
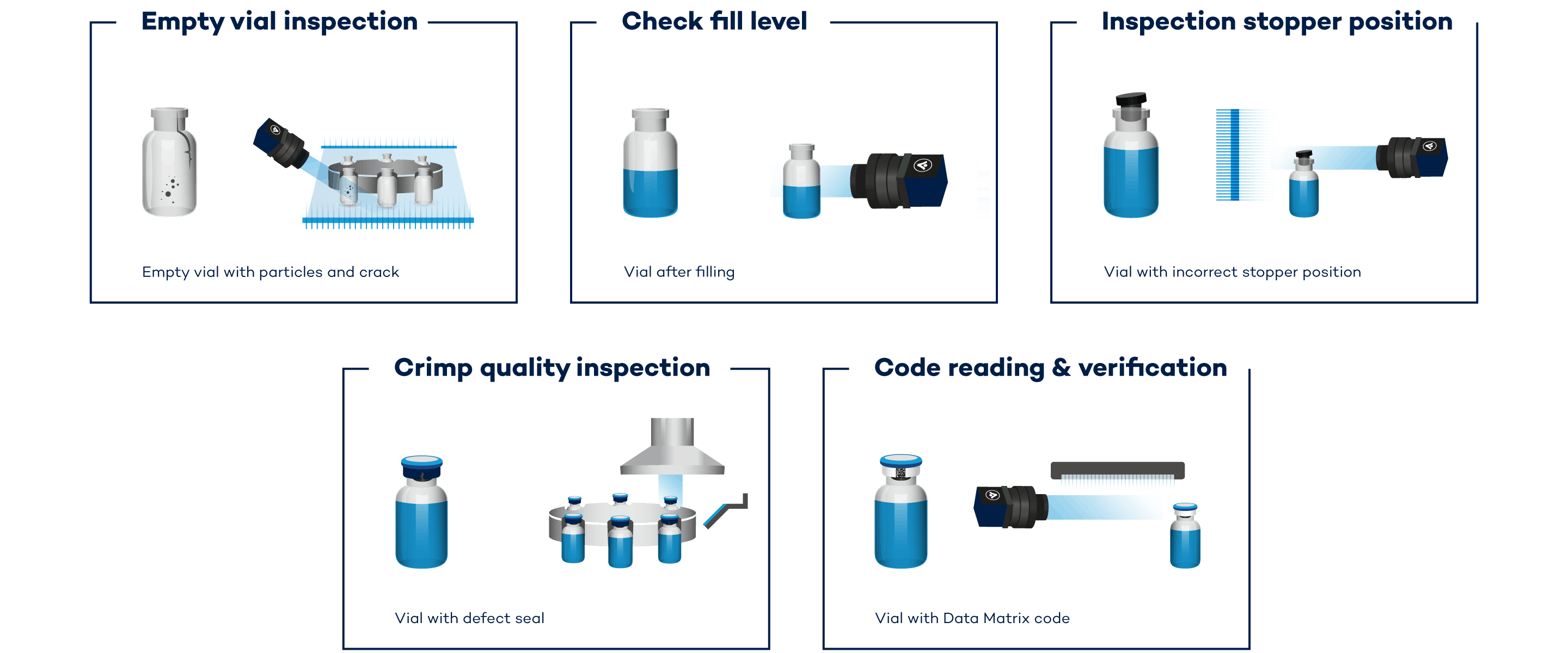
Quality Inspection for Vials During the Filling and Sealing Process
The VINSPEC HEALTHCARE inspection system covers all inspection requirements that apply to the filling and sealing of vials—inline, at filling speed. As a result, you can quickly detect unwanted process changes, correct the relevant parameter, and ensure high quality output

Crimp Caps
Is the seal sitting correctly and is the crimping complete and precise?The complete inspection process is handled by the inspection solution., which is compact and easily integrated. Mechanical rotation of the vials is not required for the 360° inspection with a single camera. A quick-change mirror set enables fast reconfiguration, saving you lots of time if frequent format changes are required.
Inspection range:
• Crimping of the sealing cap
• Defects on the cap skirt
• Height of stopper
• Color and quality of the cap lid
• Cosmetic defects on the cap and cap lid
Qualification Package – Play It Safe
Complete Vial Inspection from one source
VINSPEC HEALTHCARE from VITRONIC offers an integrated solution for automated quality inspection of vials. Thanks to its modular design, the inspection system covers a wide range of inspections along the filling process chain. Depending on requirements, one, several, or all of the individual inspections can be used.